32 volt boater
Solar Enthusiast
- Joined
- Aug 7, 2022
- Messages
- 427
+1 for nile
be sure to check out the other channel
nileblue
I am going now. I need a science fix.
Wait just found this
+1 for nile
be sure to check out the other channel
nileblue
Wow you've done a lot since I've last read your ZnBr thread.
With regards to dendrites two ideas come to mind, Redflow does %100 depth of discharge cycles to help prevent dendrites.
Another is a more rigid barrier. perhaps a neutral plate between the cathode/anode would force the dendrites to grow around the obstacle, similar to the backplate idea. or a porous ceramic material (that's obtainable) I haven't found anything yet.
I wonder if just stopping every 50 cycles and fully discharging the cell to 0v could help effectively prevent long-term dendrite growth (its a lot simpler then additives).
https://www.sciencedirect.com/science/article/pii/S2666386422000947 - i have zero idea if this would survive the WIS electrolyte environment.
I do love this back-plating idea. super curious.
Best of luck, i cant wait to see your results and thanks for taking the time to share your results with the community.
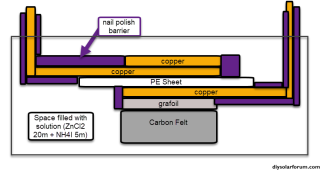

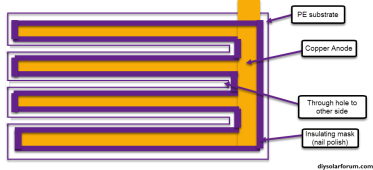
What color nail polish works best? Wait, that wasn't my question....I have done some first tests using the following architecture:
View attachment 171505
Basically strips of copper tape are first placed on a square of PE sheet, then a copper tape disc (sanded to a clean surface) and grafoil strip are placed on the anode and cathode sides respectively. A carbon felt is then glued to the grafoil's corners to secure it in place, the copper tape disc adheres using its adhesive. All exposed conductor surfaces are then insulated using nail polish to prevent any electrochemical behavior except for on the carbon felt and the copper disc anode. I used a 20m ZnCl2 + 5m NH4I solution (I ran out of potassium iodide, so I used ammonium iodide for the test). This is a Water-in-salt electrolyte (WISE). The setup is fully immersed in around 2mL of the solution.
As you can see, the above geometry ensures the Anode doesn't face the cathode and there are no direct paths for dendrites to cause a short. They would need to grow all around the electrolyte to achieve that.
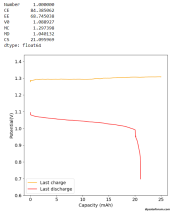
I did a couple of cycles at a current density of around 2mA/cm2, charging to 0.5mAh and discharging to 0.7V. The results are promising:
View attachment 171521
The Coulomb efficiency is quite high, above 90%, so we definitely do not get any major soluble triiodide formation in the electrolyte. All the iodide seems to plate as elemental iodide in the carbon felt (in agreement with published WISE results using KI+ZnCl2).
The loss in energy efficiency from the distance between the electrodes also seems to be limited, thanks to the high conductivity of the WISE electrolyte, even though the distance from anode to cathode is quite large as you need to go all around the PE sheet. Plating on the copper electrode looks very regular, at least from a first glance. There aren't any discernable dendrites at this state of charge (SOC). Note that higher EE value are likely achievable when charging at lower current density.
Note that the theoretical energy density you could get from this electrolyte is around 134 Ah/L (around 160 Wh/L at 1.2V) . For reference lithium iron phosphate is normally at around 220 Wh/L. The volume of the cathode material used is 0.26mL, so I would expect the cathode to hold 35mAh at a max.
A battery with this type of backplated configuration cannot be easily rolled, so likely instead of going for very fine separators and high voltage efficiencies a battery like this would work more like a lead acid battery, with larger cells stacked in rows. Basically arrays like the one I showed above stacked together to fill a box with the electrolyte then poured over to fill all the space and fill all the felt (which might require some vacuum to fully achieve wetting of the entire all the surface area.
I am now cycling this battery to 25mAh, which will take a long time per cycle, but I just want to see if it reaches the capacity and how dendrites behave at high SOC.
I agree it's an unfortunate downside, Im extremely excited to hear that the EE is still high, WIS sounds like a winner to me.A battery with this type of backplated configuration cannot be easily rolled
So is the the graphite foil and carbon felt the membrane or the polyethylene sheet? Or do they work together and the membrane?
Did you change from matte photo paper?
What color nail polish works best? Wait, that wasn't my question....
Are both the grafoil and the carbon felt part of the cathode? Would it work the same with just one or the other?
I agree it's an unfortunate downside, Im extremely excited to hear that the EE is still high, WIS sounds like a winner to me.
It shouldn't be too hard to stack cells It would be interesting to see if the cell could have a carbon felt on both sides so that during assembly the felt creates some voids for electrolyte. long term I wonder if this stacking would remain balanced over a large number of cycles, like a lead acid. but that's really a question for way down the road.


I have made a pouch design with the above structure to test a more realistic configuration. I made it with an area of 2.5cm x 2.5cm = 6.25cm2 using the same overall structure as shown before. Anode is made of copper and cathode is carbon felt over grafoil. The pouch and insulating separator are made of PE. I also put two layers of kitchen paper over the anode to ensure it has enough spacing. I sealed the entire pouch except for a small opening I used to fill with the 20m ZnCl2 + 5m NH4I electrolyte.
This is the anode side (you can see the copper surface through the wet) kitchen paper.
View attachment 171657
Here is the cathode (you can see the carbon felt)
View attachment 171658
The cell was filled with around 4.5mL of solution, then sealed with hot silicone. The max possible capacity should be around 600mAh.
I am still running the test to 25mAh on the "proof of concept" tiny device, I'll start testing this cell as soon as the test with the small one is done.
No, the surface area of the copper anode is indeed much smaller than the surface area of the felt, by a factor of perhaps a thousand or more. The felt is a very porous conductive solid, while the copper is a simple flat surface. The difference is that the zinc can deposit over zinc while iodine isn't conductive so any iodine that deposits requires new surface area to effectively deposit without a drop in conductivity.Does the surface area of the copper anode need to match the available area of the carbon felt cathode? Maybe 1:4 to match the molar densities of the electrolyte?
Have you see this video?I don't think I will play with this backplated configuration anymore. It is too hard to make because basically any mistake in the entire assembly or any deterioration of the insulating coating means you will get dendrites that will short the battery. It is a very difficult to engineer approach because any mistake is a failure mode. Seems for Zn-I it's back to flow batteries, at least until a good way to get rid of dendrites comes to light.


